Dupilumab (Dupixent) – Eczema/asthma
Sale!$88.95 $125.03
Dupilumab (Dupixent) is a biologic injection used to treat moderate-to-severe eczema and asthma by reducing inflammation and improving skin and lung health.
- Reduces eczema symptoms: Helps control itching, redness, and flare-ups.
- Improves asthma control: Decreases frequency of severe asthma attacks.
- Non-steroidal treatment: Alternative for those who cannot tolerate corticosteroids.
- Long-term relief: Maintains skin and respiratory health with regular use.
Dupilumab (Dupixent) – Eczema/Asthma Relief
Dupilumab (Dupixent) – Eczema/asthma treats moderate-to-severe eczema and certain types of asthma effectively in patients. It works by targeting specific proteins that trigger inflammation, reducing symptoms like itching, redness, and swelling. Dupilumab helps improve skin health, control asthma attacks, and enhance respiratory function. Regular use under medical supervision promotes long-term relief, better quality of life, and effective management of chronic inflammatory conditions.
What is Dupilumab?
Dupilumab, sold under the brand name Dupixent, is a monoclonal antibody that targets key inflammatory pathways in the body. Dupilumab treats moderate-to-severe eczema and certain types of asthma, especially in patients who do not respond to conventional therapies. By blocking specific proteins, Dupilumab reduces inflammation, itching redness, and swelling, helping patients manage symptoms effectively and improve overall skin and respiratory health.
Benefits of Dupilumab (Dupixent)
– Patients inject Dupilumab (Dupixent) – Eczema/asthma subcutaneously, usually every two weeks, under a doctor’s supervision.
– Particularly – Dupilumab treats moderate-to-severe eczema and certain types of asthma, particularly in patients who do not respond to conventional therapies.
– Dupilumab focuses on patients whose previous treatments failed, providing relief when conventional therapies do not work.
– Regular use under medical supervision helps improve skin health, enhance respiratory function, and support a better overall quality of life.
 Cart is empty
Cart is empty 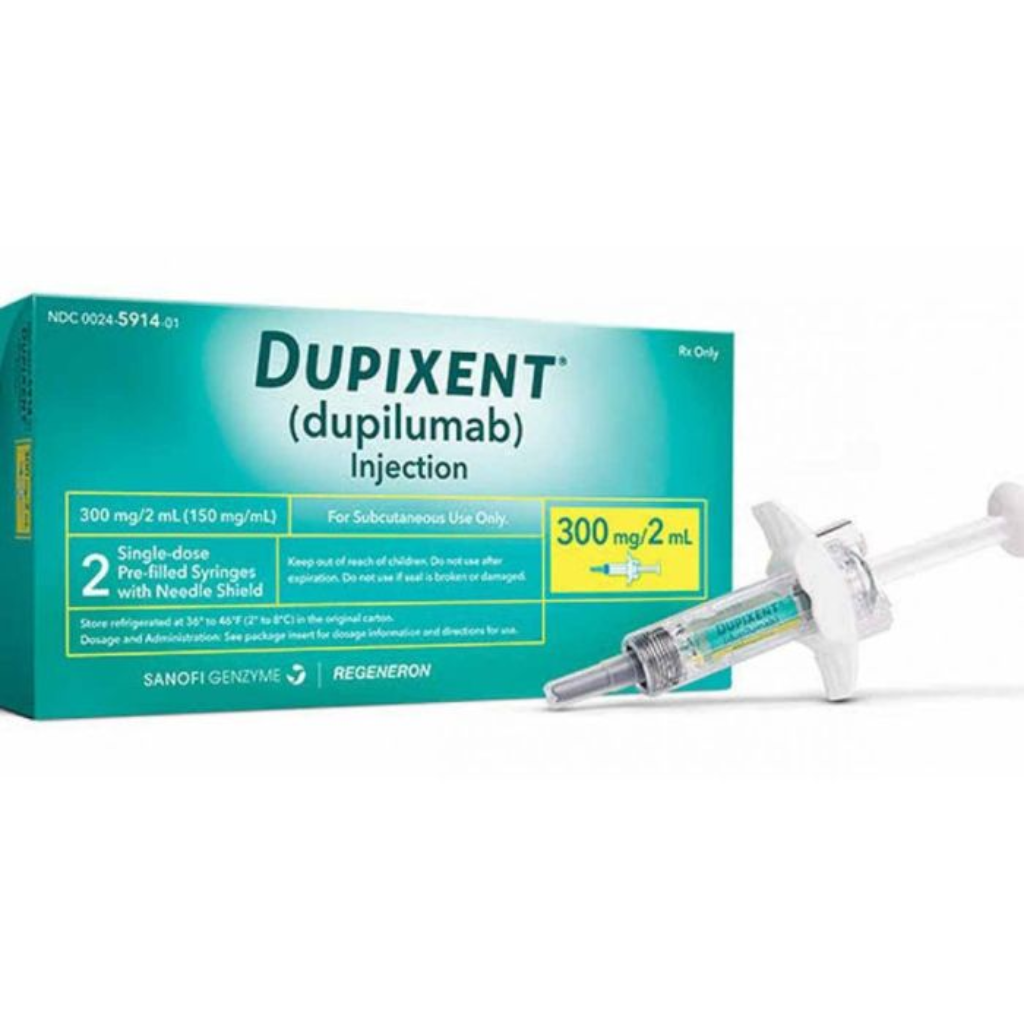

Reviews
There are no reviews yet.